The Y Chromosome Is Vanishing—But Males May Not Go With It
For over 160 million years, the Y chromosome has acted as the master trigger for male development in mammals. In most species, it carries the SRY gene, a short DNA sequence that flips the switch toward testis formation and male anatomy. Yet mounting evidence now shows that this chromosome is in a severe state of evolutionary decay. Genomic studies suggest it is slowly disappearing, raising urgent questions: what happens when the Y vanishes, and will males survive?
The surprising answer may already exist in a tiny rodent from Japan. The Amami spiny rat (Tokudaia osimensis), a critically endangered species found only on the island of Amami Ōshima, appears to have lost its Y chromosome entirely—yet it still produces healthy males. This evolutionary workaround challenges the long‑held idea that the degradation of the male chromosome inevitably leads to population collapse or extinction.
Instead, the Amami spiny rat demonstrates that the “maleness” signal can migrate from a decaying chromosome to a more stable part of the genome. This shift reveals a new chapter in how sex is determined and provides a powerful real‑world model for understanding how mammals might adapt to a post‑Y future.
How the Y Chromosome Is Dying Out
The human Y chromosome, once nearly identical in size and gene content to the X, has lost about 95% of its original genes over evolutionary time. In humans, it now carries only about 55 protein‑coding genes, down from roughly 1,438 genes originally shared with the X chromosome. This erosion is happening because the Y chromosome spends most of its life without a pairing partner.
During meiosis—the cell division that produces sperm and eggs—most chromosomes pair up and swap DNA through homologous recombination, repairing damage and reshuffling genetic variation. The Y chromosome, however, largely pairs only with the small pseudoautosomal regions of the X, leaving most of its DNA isolated. This lack of recombination makes it highly vulnerable to mutations, deletions, and the gradual loss of functional genes—a process sometimes called the “mutational trap.”
Because of this, scientists estimate that the human Y chromosome could lose its remaining functional genes and become biologically obsolete in roughly 4.6 million years, assuming the current rate of decay continues. If the Y vanishes, the standard SRY‑driven mechanism for male development disappears with it—unless the genome evolves an alternative.
The Amami Spiny Rat: A Living Post‑Y Experiment
The Amami spiny rat (Tokudaia osimensis) serves as a finished experiment in this evolutionary process. In this species, both males and females carry only a single X chromosome (XO/XO), and the Y chromosome is completely absent. Despite this, males still develop testes and produce sperm, and the species continues to reproduce.
Because the Y and the SRY gene are gone, the Amami spiny rat must use a different genetic mechanism to determine sex. This makes it an exceptional model for understanding how mammals can survive without a Y chromosome, and for probing whether similar backups could emerge in other species—including, hypothetically, humans.
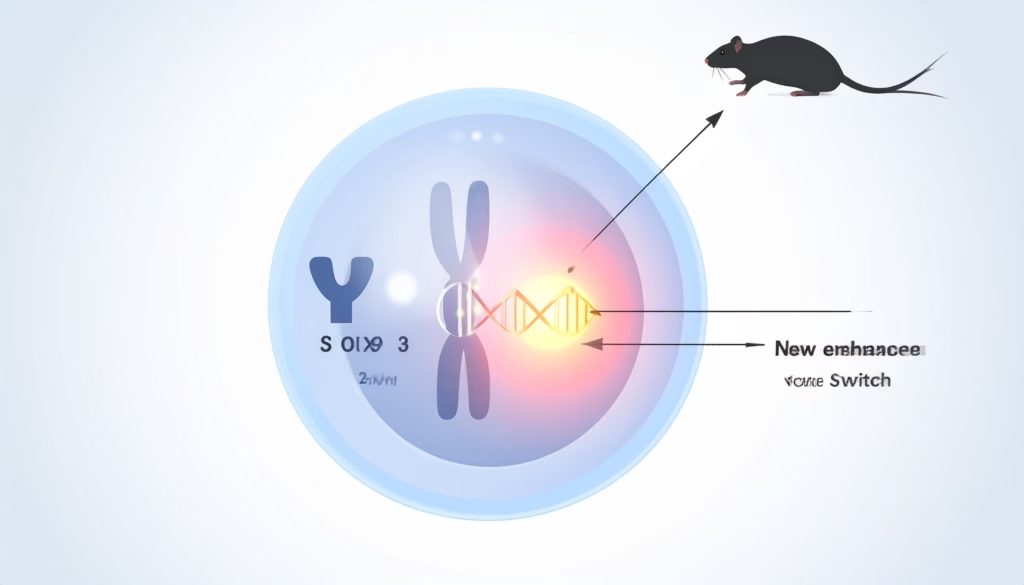
In a landmark study led by Professor Asato Kuroiwa at Hokkaido University, researchers decoded the spiny rat genome and identified the key genetic change that now regulates sex in this species. Their work, published in Proceedings of the National Academy of Sciences (PNAS), revealed that a 17,000‑base‑pair DNA duplication near the SOX9 gene on chromosome 3 has taken over the role of the male‑determining switch.
Genomic Analysis of the SOX9 Regulatory Region
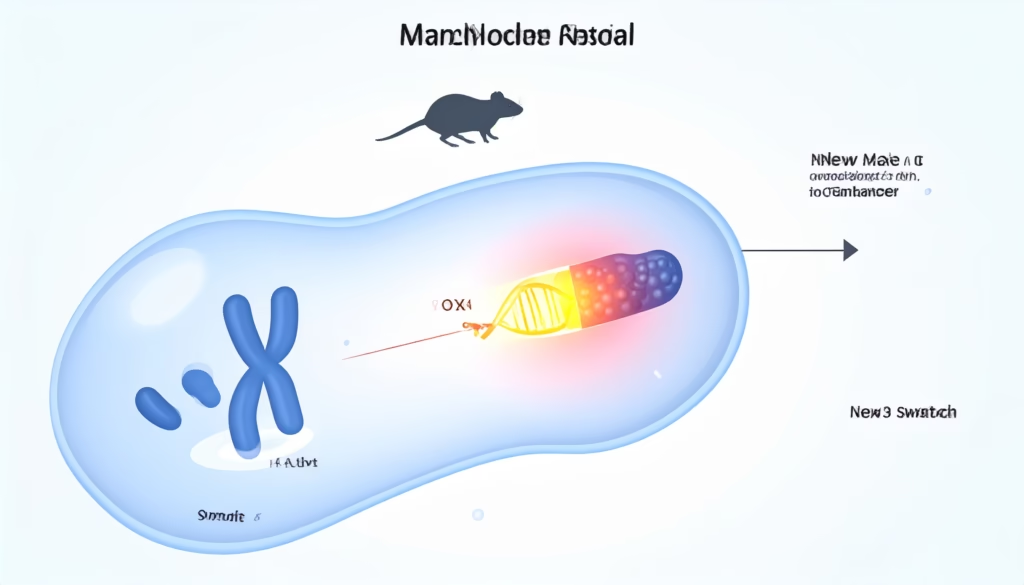
In typical mammals, the SRY gene on the Y chromosome activates the SOX9 gene, which is essential for testis development. Without SOX9 expression, undifferentiated gonads usually develop along the ovarian pathway. The PNAS study showed that in the Amami spiny rat, the SRY gene is missing, but SOX9 is still turned on in males—just through a different regulator.
By scanning the genome for sex‑specific regions, the researchers found that males carry a male‑specific tandem duplication of about 17 kilobases (kb) located upstream of the SOX9 locus on chromosome 3. This duplicated unit contains a regulatory element (enhancer) that drives SOX9 expression in the developing gonads.
The duplication is absent in females, which means that the presence or absence of this 17‑kb sequence effectively determines whether the animal develops as male or female. In functional assays, the rodent’s enhancer could activate SOX9 in mouse embryos and even shift XX mouse embryos toward male‑like gonad development, confirming its role as a new “master switch” for maleness.
Because chromosome 3 is an autosome—a non‑sex chromosome—it regularly recombines during meiosis, allowing DNA repair and making it far more stable than the Y. By moving the sex‑determining signal to this chromosome, the Amami spiny rat has effectively transplanted the male program to a more robust genomic environment.

A “Turnover Event” in Sex Chromosome Evolution
The Amami spiny rat’s new genetic switch represents what evolutionary biologists call a “turnover event” in sex chromosome evolution. In such events, a critical function—here, sex determination—shifts from one chromosome or genomic region to another while the old system fades away.
Further analysis by the Hokkaido team suggests that this new regulatory mechanism has remained stable for roughly 2 million years, indicating it is not a temporary glitch but a permanent evolutionary fix. This long‑term stability supports the idea that Y chromosome loss can be compatible with continued sexual reproduction, as long as an alternative trigger for SOX9 (or an equivalent gene) emerges.
Professor Kuroiwa summarized this insight clearly: “The Y chromosome is disappearing, but its disappearance does not mean the disappearance of males.” In other words, the concept of biological maleness appears to be more flexible than the physical chromosome that once encoded it.
Escaping the Mutational Trap: Why the Amami System Works
The Amami spiny rat’s XO/XO sex‑chromosome system helps it avoid several of the problems that plague other species losing their Y chromosomes. In many mammals, imbalances in gene dosage between the X and Y can disrupt development or cause sterility. For example, males (XY) normally have only one X chromosome, while females (XX) have two; specialized mechanisms like X‑inactivation balance this.
When the Y disappears without a replacement system, the loss of male‑specific genes can create lethal imbalances or failed sex determination. However, in the Amami spiny rat, both sexes carry a single X chromosome, and core sex‑determining information has moved to an autosome. This setup sidesteps many of the dosage problems that arise when sex chromosomes are lost or damaged in other lineages.
By anchoring the male‑determining signal on chromosome 3—a region that undergoes regular recombination—the Amami spiny rat places a crucial instruction in a part of the genome that can repair itself. This relocation effectively protects the male program from the mutational decay that defined the Y chromosome’s history.
Other Species With Y‑Free Sex Determination
The Amami spiny rat is not the only mammal to have lost its Y chromosome. The Transcaucasian mole vole (Ellobius lutescens), a rodent found in the Caucasus region, also survives without a Y chromosome, though the exact genetic mechanism differs. In the mole vole, preliminary genomic mapping suggests that variants of Sox3 or DMRT1* may play a role in sex determination, rather than relying on a SOX9 enhancer duplication like the spiny rat.
This contrast highlights an important point: mammals may have multiple backup pathways for controlling sex. These pathways can remain dormant under normal conditions but become active when the primary system—such as a Y‑linked SRY gene—is lost or damaged.
Across several species of spiny rats in the genus Tokudaia, researchers have found evidence of additional gene duplications and rearrangements associated with sex chromosomes, indicating that the transition away from the Y is not a one‑off accident but a recurring theme in evolutionary biology.
Limitations and Open Questions in Current Research
Despite the striking insights provided by the Amami spiny rat study, several limitations remain. The species is endangered and heavily protected, which has restricted the number of available tissue samples and made large‑scale population studies difficult. As a result, scientists cannot yet rule out the possibility that other subtle genetic modifiers also contribute to sex determination in this species.
Researchers also do not yet know whether the 17‑kb duplication alone is sufficient to explain male development, or whether epigenetic factors, non‑coding RNAs, or other regulatory elements play supporting roles. The long‑term genomic health of the XO/XO system—especially in terms of mutation load, reproductive efficiency, and resilience to inbreeding—is another area of active investigation.
Additionally, the exact timing and sequence of events that led to the Y chromosome’s disappearance in the Amami spiny rat lineage are still being pieced together. Studies of related Tokudaia species suggest that the evolution of these unusual sex‑chromosome systems involved a series of translocations, duplications, and structural rearrangements over millions of years.
Broader Implications for Mammalian Sex Determination
The discovery in the Amami spiny rat has broad implications for how we think about sex determination, sex chromosomes, and the future of the Y chromosome in mammals. It proves that sexual dimorphism—the existence of distinct male and female phenotypes—can be maintained even when the classic Y‑linked SRY gene is gone, as long as an alternative mechanism for turning on SOX9 (or a similar gene) evolves.
Because the new regulatory switch in the Amami spiny rat resides on an autosome, the male‑determining function is no longer tied to a fragile, non‑recombining chromosome. This relocation shields it from the mutational pressures that led to the Y’s decline in the first place.
In practical terms, the Amami spiny rat serves as a “proof‑of‑concept” for mammalian reproduction in a post‑Y‑chromosome era. It suggests that, even if the human Y chromosome continues to shrink, evolution could—if given enough time and genetic variation—move the male‑determining machinery to another genomic location.
However, such a transition would likely require many thousands of years and would not be guaranteed in every lineage. The spiny rat case also underscores the importance of protecting endangered species, as they often harbor unique genetic adaptations that can reshape our understanding of basic biology.
Future Directions and Model Potential
The Amami spiny rat’s sex‑determination mechanism is now a focal point for studies exploring how new sex‑determining genes emerge, how they interact with other developmental pathways, and how quickly such systems can evolve. Researchers are also comparing its genome with those of other Y‑deficient mammals, including additional Tokudaia species and mole voles, to identify shared patterns and lineage‑specific innovations.
These findings may also inform biomedical research. Because SOX9 plays roles beyond sex determination—including in skeletal development and certain cancers—understanding how its regulation evolves in spiny rats could yield insights into human gene regulation and disease.
In summary, the Amami spiny rat’s genetic story shows that the Y chromosome’s disappearance does not automatically spell the end of males. Instead, it illustrates how evolution can relocate, repurpose, and rewire core biological programs when the original hardware—here, the Y chromosome—begins to fail.
for more articles visit BulletinBuzzs